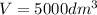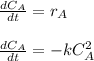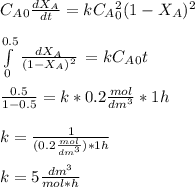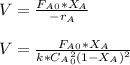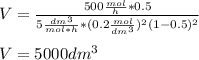Chemistry, 22.09.2020 15:01, hiyagirllyric
The elementary gas-phase reaction2A → Bis carried out in a constant-volume batch reactor where 50% conversion is achieved in 1 hour. Pure Ais charged to the reactor at an initial concentration of 0.2 mol/dm3. If the same reaction is carried outin a CSTR, what volume would be necessary to achieve 50% conversion for a feed molar flow rate of500 mol/h and an entering concentration of A of 0.2 mol/dm3? (Ans.:V = 5,000 dm3

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, adrian08022
When an object falls through the air and encounters air resistance its overall speed will be than if it had not encountered air resistance? (one word answer)
Answers: 2


Chemistry, 22.06.2019 10:30, Brookwiggington8814
What woukd most likely be the transmittance at a 0.70 m solution of solute a? a) 7.6%b) 1.1%c)4.0%d)4.6%
Answers: 1

Chemistry, 22.06.2019 12:10, coastieltp58aeg
Building glycogen from glucose molecules is an example of
Answers: 3
Do you know the correct answer?
The elementary gas-phase reaction2A → Bis carried out in a constant-volume batch reactor where 50% c...
Questions in other subjects:

Geography, 13.10.2020 15:01

Mathematics, 13.10.2020 15:01

English, 13.10.2020 15:01




Mathematics, 13.10.2020 15:01