
Chemistry, 22.09.2020 05:01, haleylayne74
A nearly flat bicycle tire becomes noticeably warmer after it has been pumped up. Approximate this process as a reversible adiabatic compression. Take the initial pressure and temperature of the air before it is put in the tire to be Pi = 1.00 bar and Ti = 298 K. The final volume of the air in the tire is Vf= 1.50 L and the final pressure is Pf = 5.00 bar. Calculate the final temperature of the air in the tire.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, erikloza12pdidtx
Which type of bond is present in hydrogen sulfide (h2s)? the table of electronegativities is given. a. hydrogen b. ionic c. nonpolar covalent d. polar covalent
Answers: 1


Chemistry, 22.06.2019 12:00, WinterStrikesBack
Solutions of sodium carbonate and silver nitrate react to form solid silver carbonate and a solution of sodium nitrate. a solution containing 3.50 g of sodium carbonate is mixed with one containing 5.00 g of silver nitrate. how many grams of sodium carbonate, silver nitrate, silver carbonate, and sodium nitrate are present after the reaction is complete?
Answers: 2

Chemistry, 22.06.2019 13:30, makenziehook8
Which is true of a liquid? it has a definite volume but not a definite mass. it has a definite mass but not a definite volume. it has a definite volume but not a definite shape. it has a definite shape but not a definite volume.
Answers: 2
Do you know the correct answer?
A nearly flat bicycle tire becomes noticeably warmer after it has been pumped up. Approximate this p...
Questions in other subjects:








Computers and Technology, 24.07.2019 17:50


Mathematics, 24.07.2019 17:50


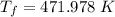
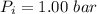
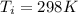
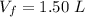
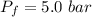
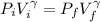
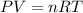
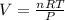
![P_i *[ \frac{n RT_i}{P_i}]^{\gamma} = P_f [ \frac{n RT_f}{P_f}]^{\gamma}](/tpl/images/0775/6605/1dfe6.png)
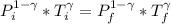
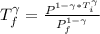
![T_f =[ \frac{ P_i ^{1 - \gamma} * T_i^{\gamma}}{P_f^{1 - \gamma}}]^{\frac{1}{\gamma} }](/tpl/images/0775/6605/333d0.png)
![T_f = T_i * [\frac{P_i}{P_f} ]^{\frac{1- \gamma}{\gamma}](/tpl/images/0775/6605/2faae.png)
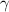 is a constant mathematically represented as
is a constant mathematically represented as 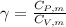
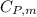 is the molar heat capacity at constant pressure
is the molar heat capacity at constant pressure 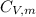 is the molar heat capacity at constant volume given as
is the molar heat capacity at constant volume given as 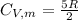
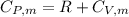
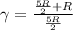
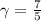
![T_f = T_i * [\frac{P_i}{P_f} ]^{\frac{1- \frac{7}{5}}{ \frac{7}{5}}](/tpl/images/0775/6605/9c28a.png)
![T_f = 298 * [\frac{1}{5} ]^{\frac{1- \frac{7}{5}}{ \frac{7}{5}}](/tpl/images/0775/6605/9e54c.png)




