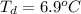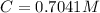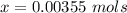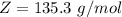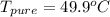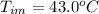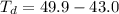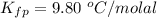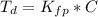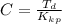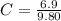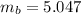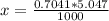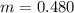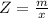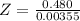Chemistry, 20.09.2020 09:01, davidleew24
Benzophenone freezing point= 49.9 C Benzophenone + unknown freezing point= 43.0 C Benzophenone mass= 5.047 g Unknown mass= .480 g 1. From the difference between the freezing points of the pure benzophenone and the unknown + benzophenone solution, calculate the freezing point depression of the solution. 2. Given the freezing point depression constant for benzophenone, Kfp= 9.80 C/molal, calculate the molality of the solution of unknown in benzophenone. (answer in m) 3. Now use the calculated value for the molality of the solution and the mass of the benzophenone to compute the number of moles of solute present in the solution. 4. Use that calculated number of moles and mass of solute to determine the approximate molar mass (Gram molecular weight) of the unknown solute. (answer in g/mol)

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:30, Riplilpeep
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1

Chemistry, 22.06.2019 13:00, yaneiryx5476
Is 9 correct? and can someone me with 10? it’s due tomorrow, you
Answers: 1
Do you know the correct answer?
Benzophenone freezing point= 49.9 C Benzophenone + unknown freezing point= 43.0 C Benzophenone mass=...
Questions in other subjects:




Computers and Technology, 12.10.2019 05:30



Biology, 12.10.2019 05:30


History, 12.10.2019 05:30