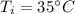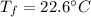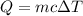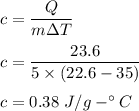Chemistry, 20.09.2020 05:01, jaymoney0531
If 5.0 g of copper cools from 35.0oC to 22.6 oC and loses 23.6 J of heat, what is the specific heat of copper? a. 0.038 J/(g. oC) b. 0.62 J/(g. oC) c. 0.076 J/(g. oC) d. 0.38 J/(g. oC)

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, clairajogriggsk
The big bang nucleosynthesis theory states that elements were produced in the first few minutes of the big bang while elements have their origins in the interiors of stars, forming much later in the history of the universe.
Answers: 1

Chemistry, 22.06.2019 10:00, aschool2000
Water's surface tension and heat storage capacity are accounted for by its a) orbitals b) weight c) hydrogen bonds d) mass e) size
Answers: 2

Chemistry, 23.06.2019 00:00, glocurlsprinces
(04.05 hc) analyze the given diagram of the carbon cycle below. part 1: which compound does c represent? part 2: name a process that could release this compound into the air. part 3: explain how the elements that form it are conserved during the carbon cycle. use complete sentences to explain your answer. justify how this compound was created from a recycling of carbon in the carbon cycle. use complete sentences to explain your answer.
Answers: 3

Chemistry, 23.06.2019 00:30, mathwiznot45
Element j is 1s 2s 2p 3s . (i) how many unpaired electrons does j have? (ii) is j a good oxidizing agent or a reducing agent? (iii) state reason for the answer.
Answers: 1
Do you know the correct answer?
If 5.0 g of copper cools from 35.0oC to 22.6 oC and loses 23.6 J of heat, what is the specific heat...
Questions in other subjects:

Mathematics, 04.05.2021 21:20

Mathematics, 04.05.2021 21:20

Mathematics, 04.05.2021 21:20

History, 04.05.2021 21:20



Mathematics, 04.05.2021 21:20