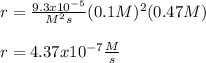The reaction 2NO(g) + O2(g) 2NO2(g) is second order in NO and first order in O2. When [NO] = 0.8 M and [O2] = 3.7 M, the observed rate of the reaction is 0.00022022 M/s. (a) What is the value of the rate constant? (d) What is the rate of reaction when [NO] = 0.1 M and [O2] = 0.47 M?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 14:00, alexisdiaz365
Your answer should have the same number or significant figures as a he starting measurement. 3201 ml =
Answers: 2

Chemistry, 22.06.2019 02:30, ulilliareinhart2
The is a particle with one unit of positive charge a. proton b. positron c. electron d. nucleus awnser quick it is a important science test!
Answers: 2

Chemistry, 22.06.2019 17:30, shookiegriffin
I'm learning about the periodic tables and what each subject's configuration is. for example, hydrogen is 1s^1, but i don't understand how you get that. can someone me understand how to figure out how to figure this out? sorry if the question makes no sense, but it would really a lot if you could me understand! you so much if you can!
Answers: 1

Chemistry, 23.06.2019 01:00, dawnparker71
Which substance—wood or silver—is the better thermal conductor? a thermal conductor is a material that requires very little heat energy to change its temperature. explain your answer.
Answers: 3
Do you know the correct answer?
The reaction 2NO(g) + O2(g) 2NO2(g) is second order in NO and first order in O2. When [NO] = 0.8 M a...
Questions in other subjects:





Social Studies, 19.01.2021 21:20



Mathematics, 19.01.2021 21:20


English, 19.01.2021 21:20


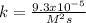
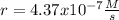
![r=k[NO]^2[O_2]](/tpl/images/0747/3983/b9fab.png)
![k=\frac{r}{[NO]^2[O_2]}\\\\k=\frac{0.00022022M/s}{(0.8M)^2(3.7M)} \\\\k=\frac{9.3x10^{-5}}{M^2s}](/tpl/images/0747/3983/c4bb6.png)