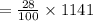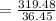Chemistry, 03.09.2020 01:01, baleycardell9901
The mass percentage of hydrochloric acid within a solution is 28.00%28.00% . Given that the density of this solution is 1.1411.141 g/mL, find the molarity of the solution.

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:00, halohero7
3. how much energy in joules is required to evaporate .0005 kg of liquid ammonia to vapor at the same temperature? 4. how much energy ( in megajoules ) is given up by .75 kg of water at 0c when it freezes to form ice at 0c? 5. explain how heat works between and at critical temperatures?
Answers: 2

Chemistry, 22.06.2019 12:10, kaitlynbernatz2778
If a molecule with a molecular formula of c13h18 is treated with an excess of h2 in the presence of finally divided pt metal under conditions required for maximum hydrogenation of the molecule to give a molecule with a formula c13h24, how many rings are in the molecule?
Answers: 3
Do you know the correct answer?
The mass percentage of hydrochloric acid within a solution is 28.00%28.00% . Given that the density...
Questions in other subjects:



History, 08.10.2019 05:20

Spanish, 08.10.2019 05:20


Mathematics, 08.10.2019 05:20

Mathematics, 08.10.2019 05:20



Mathematics, 08.10.2019 05:20