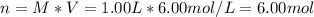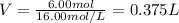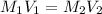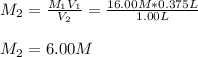Chemistry, 02.09.2020 04:01, matt199296
It is desired to make 1.00 liter of 6.00 M nitric acid from concentrated 16.00 M HNO3.A) How many moles of nitric acid are in 1.00 L of 6.00 M nitric acid?B) What volume of concentrated 16.00 M nitric acid will contain this number of moles?C) If this volume of concentrated nitric acid (answer to b) is diluted to 1.00 liter, what will be the molarity of the solution?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:40, sugardime
Choose an equation that represents an enzyme-catalyzed reaction. (a) enzyme + substrate → enzyme-substrate complex (b) enzyme + substrate ←−→ enzyme + products (c) enzyme + substrate ←−→ enzyme-substrate complex → enzyme + products (d) enzyme + substrate ←−→ enzyme-substrate complex → enzyme-substrate complex + products
Answers: 2


Chemistry, 23.06.2019 00:30, terryg4397
Fred is studying a substance that is made out of only one element. this means that
Answers: 1

Chemistry, 23.06.2019 08:30, elijah4723
If you had to research a particular disease or area of concern in veterinary medicine and science, which one would you choose? why?
Answers: 1
Do you know the correct answer?
It is desired to make 1.00 liter of 6.00 M nitric acid from concentrated 16.00 M HNO3.A) How many mo...
Questions in other subjects:

Mathematics, 27.10.2020 05:20


Mathematics, 27.10.2020 05:20



Mathematics, 27.10.2020 05:20

History, 27.10.2020 05:20