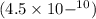Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:00, kathleendthomas
Look at the reaction below: ca(hco3)2 --> caco3 + co2 + h2o first, balance the reaction. once balanced, use dimensional analysis or another method to find out how many moles of carbon dioxide will be produced if we start with 16.5 moles of calcium bicarbonate (calcium hydrogen carbonate). = mol of co2 number needs to be reported to three significant figures.
Answers: 1


Chemistry, 21.06.2019 18:20, datboyjulio21
Complete the table for ion charge based upon their losing or gaining electrons in the outer shell. (use the periodic table as necessary.) group most likely ionic charge # of valence electrons i +1 ii +2 iii +3 iv +4 or -4 v -3 vi -2 vii -1 viii 0
Answers: 2

Chemistry, 22.06.2019 14:30, cxttiemsp021
Calculate the mass of carbon in 97.0 g of sucrose c12h22o11
Answers: 3
Do you know the correct answer?
Convert 0.00000000045 to scientific notation....
Questions in other subjects:

Biology, 07.07.2019 22:30

History, 07.07.2019 22:30


Biology, 07.07.2019 22:30

History, 07.07.2019 22:30

Mathematics, 07.07.2019 22:30


Mathematics, 07.07.2019 22:30

Mathematics, 07.07.2019 22:30

Health, 07.07.2019 22:30