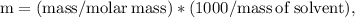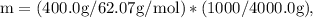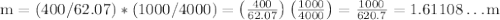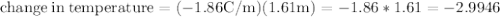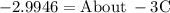Chemistry, 26.08.2020 23:01, keigleyhannah30
Ethylene glycol (C2H6O2) is used as an antifreeze in cars. If 400 g of ethylene glycol is added to 4.00 kg of water, what is the molality? Calculate how much the freezing point of water will be lowered. The freezing-point depression constant for water is Kf = –1.86°C/m.

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 23.06.2019 05:00, lifeoflashay1659
He nucleus contains the cells genetic material in the form of dna. dna is organized into our chromosomes, which are made up of thousands of that determine our traits.
Answers: 1

Chemistry, 23.06.2019 06:30, amylumey2005
The polarity of an oxygen-hydrogen bond is higher than the polarity of a nitrogen-hydrogen bond, allowing amines to be more soluble than alcohols.
Answers: 3

Chemistry, 23.06.2019 07:00, Bassoonist
How does science use models to gain a better understanding of concepts?
Answers: 1
Do you know the correct answer?
Ethylene glycol (C2H6O2) is used as an antifreeze in cars. If 400 g of ethylene glycol is added to 4...
Questions in other subjects:


History, 10.11.2020 23:00






History, 10.11.2020 23:00


Chemistry, 10.11.2020 23:00