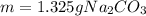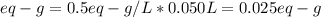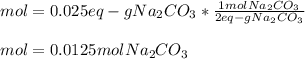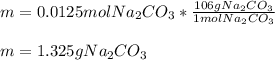Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 15:30, lovebaeforlife351
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins. co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1

Chemistry, 22.06.2019 15:40, alleshia2007
Use the periodic table to complete this equation that represents nuclear fission processesun - ba c 3 n
Answers: 2
Do you know the correct answer?
1) What mass of Na2CO3 is required to make 50cc of its seminormal solution?...
Questions in other subjects:

Mathematics, 02.03.2021 18:20

Mathematics, 02.03.2021 18:20


Biology, 02.03.2021 18:20

English, 02.03.2021 18:20

Mathematics, 02.03.2021 18:20