Chemistry, 25.08.2020 23:01, adaneri1234
g What is the half-life for a particular reaction if the rate law is rate = (1291 M⁻¹*min⁻¹)[A]² and the initial concentration of A is 0.250 M?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, KarenH3512
Use the drop-down menus to answer each question. which runner finished the 100 m race in the least amount of time? which runner stopped running for a few seconds during the race? at what distance did anastasia overtake chloe in the race?
Answers: 1

Chemistry, 22.06.2019 17:00, smelcher3900
According to the kinetic-molecular theory, what happens to a liquid when it is transferred from one container to another? the volume and the shape stay the same. the volume increases to fill the new container, but the shape stays the same. the volume stays the same, but the shape changes to fit the new container. the volume and the shape change to fill the new container.
Answers: 2
Do you know the correct answer?
g What is the half-life for a particular reaction if the rate law is rate = (1291 M⁻¹*min⁻¹)[A]² and...
Questions in other subjects:

SAT, 11.03.2021 01:00

History, 11.03.2021 01:00


Chemistry, 11.03.2021 01:00




History, 11.03.2021 01:00

Business, 11.03.2021 01:00


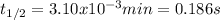
![t_{1/2}=\frac{1}{k[A]_0}](/tpl/images/0729/2407/6693f.png)