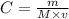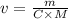Chemistry, 23.08.2020 17:01, matthewquattlebum
What volume of 1.25 M sodium hydroxide (NaOH) solution can be prepared using 60.0 g of sodium hydroxide? (The mass of one mole of NaOH is 40.0 g.)
0.667 L
0.833 L
1.20 L
1.88L

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:50, britotellerialuis
Evaluate this exponential expression,8. (2 + 3)2 – 42
Answers: 3

Chemistry, 21.06.2019 21:00, bakoeboo
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2
Do you know the correct answer?
What volume of 1.25 M sodium hydroxide (NaOH) solution can be prepared using 60.0 g of sodium hydrox...
Questions in other subjects:

Mathematics, 17.10.2021 14:00



Mathematics, 17.10.2021 14:00



History, 17.10.2021 14:00

Mathematics, 17.10.2021 14:00

Mathematics, 17.10.2021 14:00

Mathematics, 17.10.2021 14:00