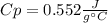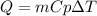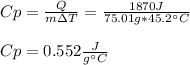Chemistry, 20.08.2020 03:01, blackbetty79
What is the specific heat of a 75.01 g piece of an unknown metal that exhibits a 45.2°C temperature change upon absorbing 1870 J of heat?

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 00:00, annsmith66
What is the result of multiplying (2.5 × 1010) × (2.0 × 10-7)? a. 5.0 × 103 b. 5.0 × 10-3 c. 5.0 × 1017 d. 5.0 × 10-17
Answers: 1

Chemistry, 22.06.2019 10:10, babyphoraaaaa
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate, m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3

Chemistry, 22.06.2019 12:40, carebear60
Quiz1. which physical state of nitrogen has the highest entropy? a solid© b gasoc liquid
Answers: 1
Do you know the correct answer?
What is the specific heat of a 75.01 g piece of an unknown metal that exhibits a 45.2°C temperature...
Questions in other subjects:


Mathematics, 06.01.2021 02:10


Chemistry, 06.01.2021 02:10

Biology, 06.01.2021 02:10

Arts, 06.01.2021 02:10