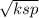Chemistry, 20.08.2020 01:01, duartealondra45
Calculate the solubility of BaCO3 (a) in pure water and (b) in a solution in which [CO32-] = 0.289 M. Solubility in pure water = M Solubility in 0.289 M CO32- = M

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:00, shayneseaton
The tendency of water molecules to stick together is referred to as a) adhesion b) polarity c) cohesion d) transpiration e) evaporation
Answers: 1


Chemistry, 22.06.2019 21:50, SoccerAllStar2
Liquid from a brewery fermentation contains 10% ethanol and 90% water. part of the fermentation product (50,000 kg/h) is pumped to a distillation column on the factory site. under current operating conditions, a distillate of 45% ethanol and 55% water is produced from the top of the column at a rate of one-tenth that of the feed. what is the composition of the waste "bottoms" from the still?
Answers: 2
Do you know the correct answer?
Calculate the solubility of BaCO3 (a) in pure water and (b) in a solution in which [CO32-] = 0.289 M...
Questions in other subjects:

Mathematics, 11.06.2020 11:57

English, 11.06.2020 11:57


Biology, 11.06.2020 11:57



Social Studies, 11.06.2020 11:57

Business, 11.06.2020 11:57



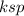 (i.e solubility constant ) for BaCO₃= 2.0 × 10⁻⁹
(i.e solubility constant ) for BaCO₃= 2.0 × 10⁻⁹