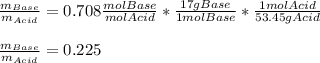Chemistry, 19.08.2020 04:01, 4300252063
Which of the following buffer systems would be the best choice to create a buffer with pH 9.10?
a) HF/KF (pKa = 3.14)
HNO2/KNO2 (pKa = 3.39)
NH3/NH4Cl (pKa = 9.25)
HClO/KClO (pKa = 7.46)
b) for the best buffer system, calculate the ratio of the molarities of the buffer components required to make the buffer
c) for the best buffer system, calculate the ratio of the masses of the buffer components required to make 1.00 L of the buffer

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 20:30, jaydenbrock
Identify the correct mole ratio for each substance. sodium chloride (nacl) na: cl = 1: ammonium nitrate (nhno) h: o = 4:
Answers: 1


Chemistry, 23.06.2019 09:30, sharmadaman641
What is the best describtion of the side of the moon that faces earth?
Answers: 2
Do you know the correct answer?
Which of the following buffer systems would be the best choice to create a buffer with pH 9.10?
a)...
Questions in other subjects:



Spanish, 08.01.2021 17:20


Mathematics, 08.01.2021 17:20

Social Studies, 08.01.2021 17:20

Engineering, 08.01.2021 17:20



Social Studies, 08.01.2021 17:20


![\frac{[Base]}{[Acid]} =0.708](/tpl/images/0724/4469/9fb67.png)
![pH=pKa+log(\frac{[Base]}{[Acid]} )\\\\log(\frac{[Base]}{[Acid]} )=9.10-9.25=-0.15\\\\\frac{[Base]}{[Acid]} =10^{-0.15}\\\\\frac{[Base]}{[Acid]} =0.708](/tpl/images/0724/4469/cfc1a.png)