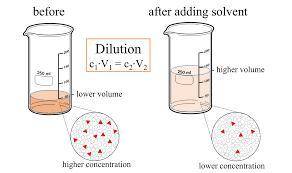
Chemistry, 19.08.2020 03:01, rwallacejr803
What is the concentration in %m/v of a 0.617 M aqueous solution of methanol (MM = 32.04 g/mol)?

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 03:00, Dkhaurithompson
Zoe is investigating the composition of substance a, an unknown substance. using chemical processes, she analyzes substance a and determines it is composed of sodium, oxygen, and hydrogen atoms in a ratio of 1 : 1 : 1. what is substance a? a. a compound b. an element c. a heterogeneous mixture d. a homogeneous mixture
Answers: 1

Chemistry, 22.06.2019 12:30, johnsont8377
Avariable that is not being directly tested during an experiment should be changed varied experimented controlled
Answers: 1

Chemistry, 22.06.2019 21:30, imalexiscv
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1
Do you know the correct answer?
What is the concentration in %m/v of a 0.617 M aqueous solution of methanol (MM = 32.04 g/mol)?...
Questions in other subjects:



Mathematics, 19.03.2020 19:23

Biology, 19.03.2020 19:23

Biology, 19.03.2020 19:23

Mathematics, 19.03.2020 19:23




Mathematics, 19.03.2020 19:24