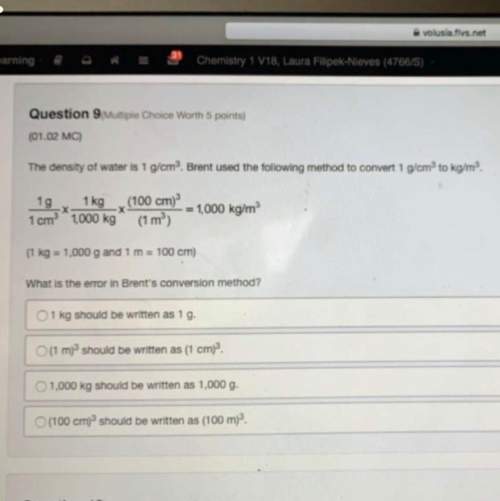
For any galvanic cell to operate at standard conditions,
a) ΔG° and ΔE° are both positive
b)...

Chemistry, 18.08.2020 21:01, wcwheelsp26w7n
For any galvanic cell to operate at standard conditions,
a) ΔG° and ΔE° are both positive
b) ΔG° and ΔE° are both negative
c) ΔG° is positive and ΔE° is negative
d) ΔG° is negative and ΔE° is positive

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 23:00, lufung8627
Consider the reaction: 2al(s) + fe2o3(s) → al2o3(s) + 2fe(s) the δhf for fe2o3(s) = -824.3 kj/mole. the δhf for al2o3(s) = -1675.7 kj/mole. finish the equation. δhrxn = [(1)( kj/mole) + (2)( kj/mole)] - [(1)( kj/mole) + (2) ( kj/mole)]
Answers: 1

Chemistry, 23.06.2019 00:00, sanaiajohnson56
In an exothermic reaction, energy may be released to the surroundings in the form of question 4 options: heat light thermal all of the above
Answers: 3

Chemistry, 23.06.2019 10:00, anonymous176
An uncovered pot of water lies out in the sun. which statements correctly describe what happens at the surface of the liquid water? 1. the vapor pressure remains constant regardless of the water temperature. 2. the vapor pressure is produced by water molecules that have evaporated. 3. the vapor pressure increases as the sun heats the water in the pot. 4. evaporation stops once the vapor pressure reaches a certain point. 5. evaporation and condensation both occur on the liquid’s surface.
Answers: 3
Do you know the correct answer?
Questions in other subjects:






History, 02.09.2019 05:50

Chemistry, 02.09.2019 05:50


English, 02.09.2019 05:50

Social Studies, 02.09.2019 05:50