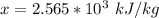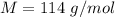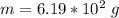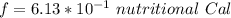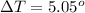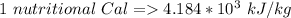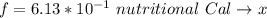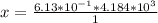A 0.0447−mol sample of a nutrient substance, with a formula weight of 114 g/mol, is burned in a bomb calorimeter containing 6.19 × 102 g H2O. Given that the fuel value is 6.13 × 10−1 in nutritional Cal when the temperature of the water is increased by 5.05°C, what is the fuel value in kJ in scientific notation?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:30, kingbot350
How many grams of magnesium metal will react completely with 8.3 liters of 5.5m hcl? show all work
Answers: 1

Chemistry, 22.06.2019 22:30, medinajocelyn45
Which compound most likely has the greatest bond energy?
Answers: 2


Chemistry, 23.06.2019 01:00, Angelofpink1143
If i had 2 m naoh solution, what does the 2 m stand for? 2 molar, but 2 of a solute in 1
Answers: 1
Do you know the correct answer?
A 0.0447−mol sample of a nutrient substance, with a formula weight of 114 g/mol, is burned in a bomb...
Questions in other subjects:

Business, 19.01.2021 21:20




Mathematics, 19.01.2021 21:20

Chemistry, 19.01.2021 21:20


Biology, 19.01.2021 21:20