
Chemistry, 12.08.2020 08:01, Homepage10
Aqueous ammonia is added to a mixture of silver chloride and water. Given that Kf for the reaction between Ag+ and NH3 is large, which of the following are true?
A) The free ions are favored over the complex ion.
B) The complex ion is favored over solid silver chloride.
C) The free Ag+ ion is unstable.
D) More silver chloride will precipitate.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:40, gabrielolivas59
The difference between the atomic number of an element and the element’s atomic mass is the number of ions.
Answers: 3

Chemistry, 22.06.2019 03:30, jabper5522
At a temperature of 393 k, the temperature of a sample of nitrogen is 1.07 atm what will the pressure be at a temperature of 478 k
Answers: 1


Chemistry, 22.06.2019 16:30, Eddie997
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2
Do you know the correct answer?
Aqueous ammonia is added to a mixture of silver chloride and water. Given that Kf for the reaction b...
Questions in other subjects:


Geography, 04.10.2021 22:20

English, 04.10.2021 22:30

Mathematics, 04.10.2021 22:30

SAT, 04.10.2021 22:30




Business, 04.10.2021 22:30


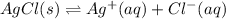
![Ag^+(aq)+2NH_3(aq)\rightleftharpoons [Ag(NH_3)_2]^+(aq)](/tpl/images/0720/0283/481b4.png)






