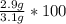Chemistry, 12.08.2020 07:01, kokokakahi
A student carries out the precipitation reaction shown below, starting with 0.030 moles of calcium nitrate. The final mass of the precipitate is 2.9 g. Answer the questions below to determine the percent yield. 3Ca(NO3)2(aq) + 2Na3PO4(aq) → Ca3(PO4)2(s) + 6NaNO3(aq) 1. a. Which product is the precipitate? b. How many moles of the precipitate would one expect to be produced from 0.030 moles of calcium nitrate? c. How many grams of solid do you expect to be produced? d. What is the percent yield?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, jocelynmarquillo1
Acamcorder has a power rating of 17 watts. if the output voltage from its battery is 7 volts, what current does it use?units:
Answers: 1

Chemistry, 22.06.2019 14:00, cheyennemitchel238
Calculate the frequency of a wave in a spring toy. the wave has a speed of 1.1 meters per second and a wavelength of 0.1 meters. *
Answers: 2

Chemistry, 22.06.2019 15:30, christopherluckey7
The reactions of photosynthesis occur in the of plant cell? a. mitochondria. b. lysosomes. c. chloroplasts. d. chlorophyll
Answers: 1
Do you know the correct answer?
A student carries out the precipitation reaction shown below, starting with 0.030 moles of calcium n...
Questions in other subjects:

Mathematics, 19.01.2021 17:00

Mathematics, 19.01.2021 17:00

Mathematics, 19.01.2021 17:00

Biology, 19.01.2021 17:00

Chemistry, 19.01.2021 17:00


Mathematics, 19.01.2021 17:00

English, 19.01.2021 17:00

Arts, 19.01.2021 17:00

Chemistry, 19.01.2021 17:00