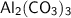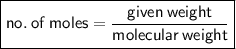Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 14:10, alanflores40
Amonoprotic acid is an acid that donates a single proton to the solution. suppose you have 0.140 g of a monoprotic acid dissolved in 35.0 ml of water. this solution is then neutralized with 14.5 ml of 0.110 m naoh. what is the molar mass of the acid?
Answers: 1

Chemistry, 21.06.2019 18:30, jadepotts3965
Calculate the change in entropy if br2(l) is converted into gaseous br atoms. s° for br2(l) = 152.2 j/(mol•k) s° for br2(g) = 245.5 j/(mol•k) s° for br(g) = 175.0 j/(mol•k)
Answers: 2


Chemistry, 22.06.2019 05:20, barry14201
Identify and describe the three ways that mutations affect organisms.
Answers: 1
Do you know the correct answer?
Calculate the number of moles equivalent 10.0 gram of aluminium carbonate...
Questions in other subjects:

Physics, 05.05.2021 04:30

Mathematics, 05.05.2021 04:30



History, 05.05.2021 04:30

History, 05.05.2021 04:30



History, 05.05.2021 04:30