
For the set of ionic compounds, LiCl, AgCl, PbCl2, choose the correct characterization of their solubilities in water from the response list. Group of answer choices All three salts are soluble. None of the three salts are soluble. Two of the three salts are soluble. One of the three salts is soluble.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 14:00, rosetoheart2
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 2

Chemistry, 22.06.2019 16:00, sassy11111515
The chemical equation below shows the reaction of sodium (na) and chlorine (cl) to form sodium chloride (nacl). 2na + cl2 → 2nacl in this equation, which of the following is a reactant? i. sodium ii. chlorine iii. sodium chloride
Answers: 1


Chemistry, 23.06.2019 10:30, villarrealc1987
Using the periodic table, complete the following. element: hydrogen symbol: h₂ molecular weight: g mass of one mole: g/mol
Answers: 3
Do you know the correct answer?
For the set of ionic compounds, LiCl, AgCl, PbCl2, choose the correct characterization of their solu...
Questions in other subjects:

Health, 04.07.2019 15:00


Social Studies, 04.07.2019 15:00

Mathematics, 04.07.2019 15:00


Mathematics, 04.07.2019 15:00

Physics, 04.07.2019 15:00

Social Studies, 04.07.2019 15:00

Arts, 04.07.2019 15:00


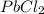 would be insoluble in cold water while LiCl would be soluble.
would be insoluble in cold water while LiCl would be soluble.



