Chemistry, 12.08.2020 06:01, iamabeast51
A chemist fills a reaction vessel with 0.978 g aluminum hydroxide AlOH3 solid, 0.607 M aluminum Al+3 aqueous solution, and 0.396 M hydroxide OH− aqueous solution at a temperature of 25.0°C.
Under these conditions, calculate the reaction free energy ΔG for the following chemical reaction:
Al(OH)3(s) = A1+ (aq) +30H (aq)
Use the thermodynamic information in the ALEKS Data tab. Round your answer to the nearest kilojoule.
KJ

Answers: 2
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 12:30, azzyla2003
Write the chemical formula for a compound that is made of an element from group 1 and an element from group 17
Answers: 1

Chemistry, 22.06.2019 14:00, BrandyLeach01
How does the presence of oxygen affect the chemical pathways used to extract energy from glucose?
Answers: 3
Do you know the correct answer?
A chemist fills a reaction vessel with 0.978 g aluminum hydroxide AlOH3 solid, 0.607 M aluminum Al+3...
Questions in other subjects:

Mathematics, 27.06.2019 23:30


History, 27.06.2019 23:30

Social Studies, 27.06.2019 23:30

History, 27.06.2019 23:30


Mathematics, 27.06.2019 23:30



Chemistry, 27.06.2019 23:30


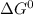 = 168.12 kJ
= 168.12 kJ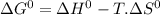
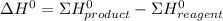
![\Delta H^{0} = [3*(-299.9)+(-524.7)] - (-1277)](/tpl/images/0719/2708/1f302.png)
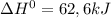
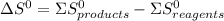
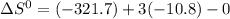
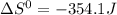
![\Delta G^{0} = 62600 - [298.(-354.1)]](/tpl/images/0719/2708/9ef3e.png)