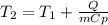Chemistry, 12.08.2020 07:01, FlowerChild1229
A 135 g sample of H20 at 85°C is cooled. The water loses a total of 15 kJ of energy in the cooling
process. What is the final temperature of the water? The specific heat of water is 4.184 J/g.°C.
A. 112°C
B. 58°C
C. 70°C
D. 84°C
E. 27°C

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:30, janetexcoelho
What does the mass of 0.7891 mol of ferric oxide (fe2o3)
Answers: 1


Chemistry, 22.06.2019 16:30, danbelucio
Explain in detail of the four major scientific developments that spurred the formulation of the plate tectonics theory
Answers: 2

Chemistry, 22.06.2019 17:10, gungamer720
Increasing the substrate concentration in an enzymatic reaction could overcome which of the following? a) the need for a coenzymeb) allosteric inhibitionc) competitive inhibitiond) insufficient cofactors
Answers: 1
Do you know the correct answer?
A 135 g sample of H20 at 85°C is cooled. The water loses a total of 15 kJ of energy in the cooling...
Questions in other subjects:



Law, 26.10.2020 16:00

Biology, 26.10.2020 16:00


Mathematics, 26.10.2020 16:00



Social Studies, 26.10.2020 16:00

Chemistry, 26.10.2020 16:00


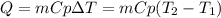
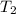 we obtain:
we obtain: