The change in the oxidation number of the iron atom in the reaction is from +3 to 0
Oxidation is simply defined as the the loss of electron. However, Oxidation number simply talks about the number of electrons that is either gained or lossed during bond formation.
The change in the oxidation number of iron in the reaction can be obtained as follow:
2Fe₂O₃(s) + 3C(s) → 4Fe(s) + 3CO₂(g)
Oxidation number of Fe in Fe₂O₃
Oxidation number of Fe₂O₃ = 0 (ground state)
Oxidation number of oxygen = –2
Oxidation number of Fe =?
Fe₂O₃ = 0
2Fe + 3O = 0
2Fe + 3(–2) = 0
2Fe – 6 = 0
Collect like term
2Fe = 6
Divide both side by 2
Fe = 6/2
Fe = +3
Thus, the oxidation number of Fe in Fe₂O₃ is +3
Oxidation number of Fe (ground state) is zero
Therefore, the change in the oxidation number of the iron, Fe, atom in the reaction is from +3 to 0
Learn more: link

















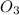 with
with 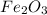 has oxidation state -6, So any atom with it would have an oxidation state of +6 to give the resultant of zero.
has oxidation state -6, So any atom with it would have an oxidation state of +6 to give the resultant of zero. 



