

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:30, cadenhuggins2
Predict whether the changes in enthalpy, entropy, and free energy will be positive or negative for the boiling of water, and explain your predictions. how does temperature affect the spontaneity of this process?
Answers: 1


Chemistry, 22.06.2019 20:20, Matseleng3775
The characteristics of two different types of reactions are shown below: reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of an element. which statement is true about the atoms of the elements that participate in the two reactions? their identity changes in both reaction a and reaction b. their identity changes in reaction a but not in reaction b. their identity changes in reaction b but not in reaction a. their identity remains the same in both reaction a and reaction b.
Answers: 1

Chemistry, 22.06.2019 22:30, COOLIOMARIS
What three things does a balanced equation show you?
Answers: 1
Do you know the correct answer?
In calculating the equilibrium constant for a reaction, the coefficients of the chemical equation ar...
Questions in other subjects:

Biology, 17.10.2021 08:40



Mathematics, 17.10.2021 08:40

English, 17.10.2021 08:40

English, 17.10.2021 08:40

Mathematics, 17.10.2021 08:40

English, 17.10.2021 08:40

Mathematics, 17.10.2021 08:40

World Languages, 17.10.2021 08:40


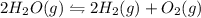
![K_{eq}=\frac{[H_2]^2[O_2]}{[H_2O]^2}](/tpl/images/0715/9643/1f40d.png)




