Chemistry, 30.07.2020 04:01, jadkins842
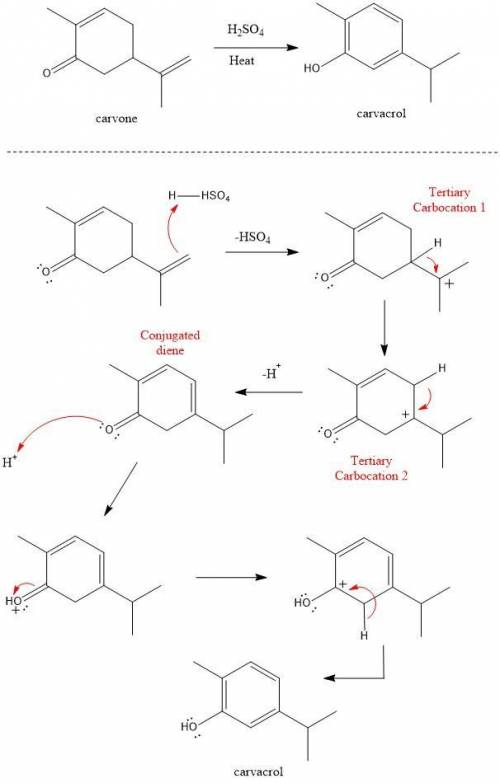
Heating carvone with aqueous sulfuric acid converts it into carvacrol. The mechanism involves the following steps:
1. The terminal alkene of carvone reacts with acid to form tertiary carbocation 1;
2. A hydride shift results in the formation of tertiary carbocation 2;
3. Deprotonation of the ring leads to conjugated diene 3;
4. Deprotonation at the α carbon leads to the product carvacrol.
Required:
Draw the mechanism and then draw the structure of tertiary carbocation 2.

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 22:30, safiyabrowne7594
[ou.03jthe pictures below show the wavelengths and intensities of electromagnetic radiations emitted by three stars, star 1, star 2, and star 3. intensity intensity- intensity- 1000 3500 6000 8500 11000 wavelength (a) star 1 1000 3500 6000 8500 11000 1000 3500 6000 8500 11000 wavelength (a) wavelength (a) star 2 star 3 which of these statements is correct about the color of the three stars? star 2 is white in color o star 2 is yellow in color star 1 and star 3 are yellow in color star 1 and star 3 are white in color
Answers: 1

Chemistry, 22.06.2019 23:40, sydneykated
The kw for water at 0 °c is 0.12× 10–14 m2. calculate the ph of a neutral aqueous solution at 0 °c.
Answers: 2
Do you know the correct answer?
Heating carvone with aqueous sulfuric acid converts it into carvacrol. The mechanism involves the fo...
Questions in other subjects:

Mathematics, 03.07.2019 14:00


Mathematics, 03.07.2019 14:00

Chemistry, 03.07.2019 14:00

History, 03.07.2019 14:00



Mathematics, 03.07.2019 14:00


Mathematics, 03.07.2019 14:00