
Chemistry, 30.07.2020 01:01, pablohc200021
What would be the voltage (Ecell) of a voltaic cell comprised of Cr (s)/Cr3+(aq) and Fe (s)/Fe2+(aq) if the concentrations of the ions in solution were [Cr3+] = 0.75 M and [Fe2+] = 0.25 M at 298K?

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 18:50, cj31150631
Question 3(multiple choice worth 4 points) (04.04 lc) what does it mean when an element is reduced? it empties a valance shell, reducing its atomic radius. it gains electrons, reducing its overall charge. it increases electronegativity, reducing its ability to bond. it loses electrons, reducing its electron number.
Answers: 1
Do you know the correct answer?
What would be the voltage (Ecell) of a voltaic cell comprised of Cr (s)/Cr3+(aq) and Fe (s)/Fe2+(aq)...
Questions in other subjects:

Mathematics, 13.10.2019 02:30


Geography, 13.10.2019 02:30




Advanced Placement (AP), 13.10.2019 02:30


English, 13.10.2019 02:30


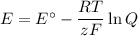
![Q = \dfrac{\text{[Fe}^{2+}]^{3}}{ \text{[Cr}^{3+}]^{2}} = \dfrac{0.25^{3}}{ 0.75^{2}} =\dfrac{0.0156}{0.562} = 0.0278\\\\E = 0.33 - \left (\dfrac{8.314 \times 298}{6 \times 96485}\right ) \ln(0.0278)\\\\=0.33 -0.00428 \times (-3.58) = 0.33 + 0.0153 = \textbf{0.35 V}\\\text{The cell potential is }\large\boxed{\textbf{0.35 V}}](/tpl/images/0715/1287/2ffcc.png)





