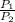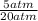Chemistry, 29.07.2020 05:01, miannasanderso8146
2) 2.5 mol of an ideal gas at 20 oC under 20 atm pressure, was expanded up to 5 atm pressure via; (a) adiabatic reversible and (b) adiabatic irreversible process. Calculate the values of w, q, ΔU, ΔH for each process. (Cv = 5 cal / mol. K ≈ 5/2 R; R ≈ 2 cal / mol. K) (Please find the desired values by making the corresponding derivations

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 18:00, jessicannoh5965
The fact that the total amount of energy in a system remains constant is a(n)
Answers: 1


Chemistry, 22.06.2019 23:00, DESI111609
What is the average rate of the reaction between 10 and 20 s?
Answers: 1
Do you know the correct answer?
2) 2.5 mol of an ideal gas at 20 oC under 20 atm pressure, was expanded up to 5 atm pressure via; (a...
Questions in other subjects:

Mathematics, 02.08.2019 14:00

Mathematics, 02.08.2019 14:00

Mathematics, 02.08.2019 14:00






Mathematics, 02.08.2019 14:00