
To determine the concentration of chloride ion (Cl-) in a 100 mL sample of ground water, a chemist adds a large enough volume of AgNO3 solution to precipitate all Cl- as AgCl. The mass of the resulting precipitate is 93.9 mg. What is the chloride ion concentration in milligrams of chloride per liter of ground water

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:00, kleathers97
Nconcentration refers to the molar concentration of an ion in solution. it may be identical to, or greater or less than, the molar concentration of the compound containing the ion that was used to make the solution. for soluble salts, the molarity of a particular ion is equal to the molarity of that compound times the subscript for that ion. for example, 1 m of alcl3 is 1 m in al3+ and 3 m in cl−. 1 m of (nh4)2so4 is 2 m in nh4+ and 1 m in so42−. part a what is the concentration of k+ in 0.15 m of k2s? view available hint(s) nothing m m part b if cacl2 is dissolved in water, what can be said about the concentration of the ca2+ ion? view available hint(s) if is dissolved in water, what can be said about the concentration of the ion? it has the same concentration as the cl− ion. its concentration is half that of the cl− ion. its concentration is twice that of the cl− ion. its concentration is one-third that of the cl− ion. part c a scientist wants to make a solution of tribasic sodium phosphate, na3po4, for a laboratory experiment. how many grams of na3po4 will be needed to produce 550 ml of a solution that has a concentration of na+ ions of 0.700 m ? express your answer numerically in grams. view available hint(s) mass of na3po4 n a 3 p o 4 = nothing g provide feedback
Answers: 3

Chemistry, 22.06.2019 21:00, alwaysneedhelp84
Write a balanced equation showing the formation of copper (ii) nitrite from its elements
Answers: 1

Chemistry, 23.06.2019 06:00, fjsdfj1284
Which change will decrease the number of effective collisions during a chemical reaction? a. adding a catalyst b. increasing the surface area c. decreasing the temperature d. increasing the reactant concentrations e. increasing the volume of the reactants
Answers: 2

Chemistry, 23.06.2019 06:10, tammydbrooks43
How much would the freezing point of water decrease if 4 mol of nacl were added to 1 kg of water (kf=1.86 degrees c/(mol/kg) for water and i=2 for nacl a- 7.44 degrees c b- 14.88 c 3.72 d 1.86
Answers: 1
Do you know the correct answer?
To determine the concentration of chloride ion (Cl-) in a 100 mL sample of ground water, a chemist a...
Questions in other subjects:

Physics, 05.10.2021 05:40

Social Studies, 05.10.2021 05:40

English, 05.10.2021 05:40


English, 05.10.2021 05:40

Mathematics, 05.10.2021 05:40

Mathematics, 05.10.2021 05:40



Mathematics, 05.10.2021 05:40


![[Cl^-]=232.3\frac{mgCl^-}{L}](/tpl/images/0713/4238/a2cee.png)
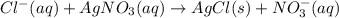
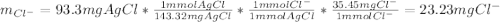
![[Cl^-]=\frac{23.2mgCl^-}{0.100L}\\](/tpl/images/0713/4238/dfd21.png)




