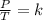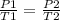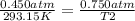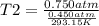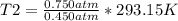Chemistry, 27.07.2020 01:01, jaidenlaine9261
. Calculate the final Celsius temperature of sulfur dioxide gas if 50.0 mL of the gas at 20 C and 0.450 atm is heated until the pressure is 0.750 atm. Assume that the volume remains constant.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:00, kkmonsterhigh18
The diagram below shows a cell placed in a solution. a cell is shown placed inside a beaker. it is labeled cell. the solution inside the beaker is labeled 40% salt solution and the solution inside the cell is labeled 20% salt solution. only water is allowed to move in and out of the cell. what will most likely happen to the cell? it will expand as water moves out of it. it will shrink as water moves out of it. it will expand as water moves into it. it will shrink as water moves into it.
Answers: 2


Chemistry, 22.06.2019 14:30, joejoefofana
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀ pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4. 0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3
Do you know the correct answer?
. Calculate the final Celsius temperature of sulfur dioxide gas if 50.0 mL of the gas at 20 C and 0....
Questions in other subjects:

Mathematics, 10.09.2020 06:01

Mathematics, 10.09.2020 06:01

Mathematics, 10.09.2020 06:01

Mathematics, 10.09.2020 06:01

Mathematics, 10.09.2020 06:01

Mathematics, 10.09.2020 06:01

Mathematics, 10.09.2020 06:01

Mathematics, 10.09.2020 06:01

Mathematics, 10.09.2020 06:01

Physics, 10.09.2020 06:01