
Chemistry, 24.07.2020 21:01, awesomegamergurl13
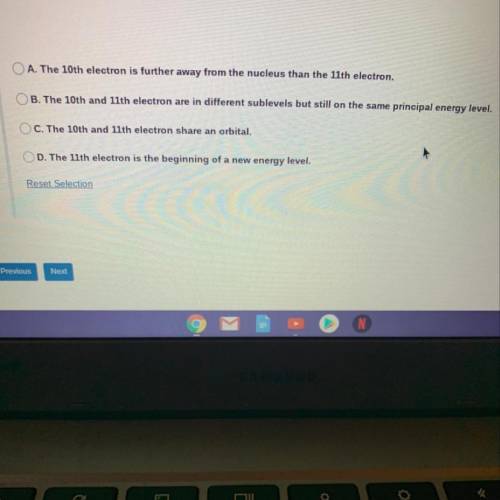
Use the graph below to answer the following question. Observe the ionization energy needed to remove the 10th and 11th electrons. What does this indicate?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, brittanysanders
When you perform this reaction, what could remain at the end of the reaction? check all that apply. excess reactant aqueous copper chloride excess reactant aluminum oxygen product solid copper carbon dioxide product aqueous aluminum chloride water
Answers: 2

Chemistry, 22.06.2019 06:00, jwood287375
When a spring is compressed, the energy changes from kinetic to potential. which best describes what is causing this change?
Answers: 3

Chemistry, 22.06.2019 10:00, valdezlizbeth6652
Why is carbon ideal for making different compounds?
Answers: 2

Chemistry, 22.06.2019 11:40, Wemaybewrong
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1
Do you know the correct answer?
Use the graph below to answer the following question. Observe the ionization energy needed to remove...
Questions in other subjects:

Mathematics, 29.10.2020 07:30

English, 29.10.2020 07:30


Mathematics, 29.10.2020 07:30

World Languages, 29.10.2020 07:30

Advanced Placement (AP), 29.10.2020 07:30

Mathematics, 29.10.2020 07:30


Mathematics, 29.10.2020 07:30

Mathematics, 29.10.2020 07:30






