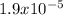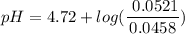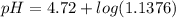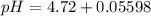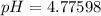Chemistry, 23.07.2020 19:01, moneyyfletcher
A 25.0-mL sample of 0.150 M hydrazoic acid, HN3, is titrated with a 0.150 M NaOH solution. What is the pH after 13.3 mL of base is added? The Ka of hydrazoic acid = 1.9 x 10-5.

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 08:00, gomezyonathan93
An observation that requires measurement is called quantitative observable or qualitative
Answers: 1

Chemistry, 22.06.2019 13:30, bryce99
In a ni-cd battery, a fully charged cell is composed of nickelic hydroxide. nickel is an element that has multiple oxidation states. assume the following proportions of the states: nickel charge proportions found 0 0.17 +2 0.3 +3 0.33 +4 0.5 (a) determine the mean of the nickel charge. enter the answer to 2 decimal places.(b) determine the cumulative distribution function of nickel charge.
Answers: 2
Do you know the correct answer?
A 25.0-mL sample of 0.150 M hydrazoic acid, HN3, is titrated with a 0.150 M NaOH solution. What is t...
Questions in other subjects:


History, 25.06.2019 09:30


Mathematics, 25.06.2019 09:30


Mathematics, 25.06.2019 09:30



Mathematics, 25.06.2019 09:30



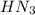 =
= 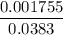 = 0.0458 M
= 0.0458 M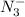 =
= 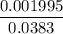 = 0.0521 M
= 0.0521 M