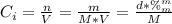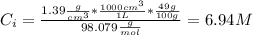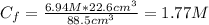Chemistry, 23.07.2020 03:01, hncriciacheichi
A solution of 49.0% H2SO4 by mass has a density of 1.39 g cm−3 at 293 K. A 22.6 cm3 sample of this solution is mixed with enough water to increase the volume of the solution to 88.5 cm3 . Find the molarity of sulfuric acid in this solution.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:00, angeljohnson2081
Which object forms when a supergiant runs out of fuel? a red giant a black hole a white dwarf a neutron star
Answers: 1

Chemistry, 22.06.2019 06:30, coreyslotte
Use examples from the article to explain one positive and one negative effect that chemistry has had on society.
Answers: 2

Chemistry, 22.06.2019 11:00, RidhaH
Which statement correctly identifies the scientific question and describes why the question is scientific? question 1 refers to the supernatural. question 2 reflects a moral or social value. question 3 refers to something that can be measured. question 4 reflects a question that can’t be observed.
Answers: 1

Chemistry, 23.06.2019 00:30, mariaramirez110379
On the periodic table, elements are arranged by which of the following. a. mass numbers. b. increasing atomic number. c. alphabetical order. or d. density
Answers: 1
Do you know the correct answer?
A solution of 49.0% H2SO4 by mass has a density of 1.39 g cm−3 at 293 K. A 22.6 cm3 sample of this s...
Questions in other subjects:

Physics, 11.10.2021 05:10

Mathematics, 11.10.2021 05:10

History, 11.10.2021 05:10

Business, 11.10.2021 05:10


Biology, 11.10.2021 05:10

English, 11.10.2021 05:10

Business, 11.10.2021 05:10

Mathematics, 11.10.2021 05:10


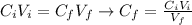
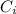 : is the initial concentration of the acid
: is the initial concentration of the acid 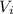 : is the initial volume of the solution = 22.6 cm³
: is the initial volume of the solution = 22.6 cm³ 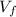 : is the final volume of the solution = 88.5 cm³
: is the final volume of the solution = 88.5 cm³