

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:10, lilyjordan5972
How do forces between particles in gases compare to forces in the other states of matter? o a. the forces in gases are stronger than forces in solids but weaker than forces in liquids. o b. the forces in gases are weaker than forces in solids but stronger than forces in liquids. o c. the forces in gases are weaker than forces in solids and liquids. o d. the forces in gases are stronger than forces in solids and liquids. submit
Answers: 1

Chemistry, 22.06.2019 00:00, chameleonsarelife
Select the correct answer. which statement is true about a polarized object? o a. it gains electrons and becomes negatively charged. ob. it gains protons and becomes positively charged. oc. the number of positive and negative charges can be the same. od. it has to be a metal. o e. there is no change in the distribution of the charge in the object. reset next what
Answers: 3

Chemistry, 22.06.2019 12:50, khorasanpublic
The number at the end of an isotope’s name is the number.
Answers: 1

Chemistry, 22.06.2019 13:50, amandamac7339
Abeaker with 2.00×102 ml of an acetic acid buffer with a ph of 5.000 is sitting on a benchtop. the total molarity of acid and conjugate base in this buffer is 0.100 m. a student adds 4.70 ml of a 0.360 m hcl solution to the beaker. how much will the ph change? the pka of acetic acid is 4.740.
Answers: 1
Do you know the correct answer?
At 100 oC, Kc=0.36. If a 1.00-L flask initially contains 0.100 M N2O4, what will be the equilibrium...
Questions in other subjects:


Mathematics, 17.11.2019 20:31

Mathematics, 17.11.2019 20:31





Mathematics, 17.11.2019 20:31


History, 17.11.2019 20:31


![[NO_2]=0.12M](/tpl/images/0710/8191/783ef.png)
![Kc=\frac{[NO_2]^2}{[N_2O_4]}](/tpl/images/0710/8191/7c0a3.png)
 (ICE methodology) we can write:
(ICE methodology) we can write:![Kc=\frac{(2x)^2}{[N_2O_4]_0-x}](/tpl/images/0710/8191/c9a45.png)
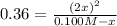
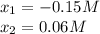
![[NO_2]=2*0.06M](/tpl/images/0710/8191/114a1.png)




