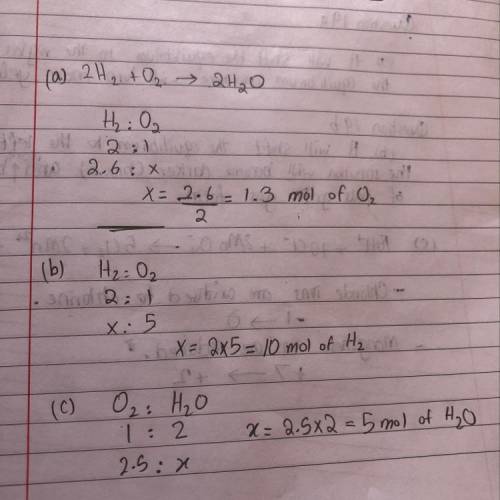The reaction of hydrogen with oxygen produces water.
2H2(g) + O2(8) - 2H2O(g)
a. How many moles of O2 are required to react with 2.6 mol
of H2?
b. How many moles of H, are needed to react with 5.0 mol
of O2?
c. How many moles of H2O form when 2.5 mol of O2
reacts?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:00, chameleonsarelife
Select the correct answer. which statement is true about a polarized object? o a. it gains electrons and becomes negatively charged. ob. it gains protons and becomes positively charged. oc. the number of positive and negative charges can be the same. od. it has to be a metal. o e. there is no change in the distribution of the charge in the object. reset next what
Answers: 3



Chemistry, 22.06.2019 01:30, lizethdominguez037
What is the value of keq for the reaction expressed in scientific notation
Answers: 1
Do you know the correct answer?
The reaction of hydrogen with oxygen produces water.
2H2(g) + O2(8) - 2H2O(g)
a. How many mol...
a. How many mol...
Questions in other subjects:

Mathematics, 04.06.2021 15:20



Mathematics, 04.06.2021 15:20

Mathematics, 04.06.2021 15:20


Social Studies, 04.06.2021 15:20


Computers and Technology, 04.06.2021 15:20

Mathematics, 04.06.2021 15:20