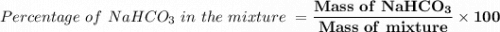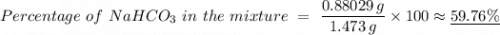A 1.555-g sample of baking soda decomposes with heat to produce 0.991 g Na2CO3. Refer to Example Exercise 14.l and show the calculation for the theoretical yield of Na2CO3.
What is the percent yield of sodium carbonate, Na2CO3?
6. A 1473-g unknown mixture with baking soda is heated and has a mass loss of 0.325 g. Refer to Example Exercise 14.2 and show the calculation for the percentage NaHCOs in the mixture.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:40, trinityanne1738
Asolid that forms and separates from a liquid mixture is called
Answers: 2


Chemistry, 22.06.2019 22:40, destineysarah
Covalent bonds generally form when the bonded elements have a difference in electronegativity less than 1.5. subtract the electronegativities for the following pairs of elements and predict whether they form a covalent bond. electronegativity difference of c and c: ionic covalent electronegativity difference of mg and cl: ionic covalent
Answers: 1
Do you know the correct answer?
A 1.555-g sample of baking soda decomposes with heat to produce 0.991 g Na2CO3. Refer to Example Exe...
Questions in other subjects:

Chemistry, 24.06.2019 17:00

Spanish, 24.06.2019 17:00

Chemistry, 24.06.2019 17:00

English, 24.06.2019 17:00

Mathematics, 24.06.2019 17:00


Mathematics, 24.06.2019 17:00

Mathematics, 24.06.2019 17:00


Mathematics, 24.06.2019 17:00


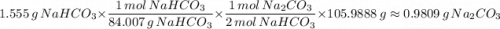
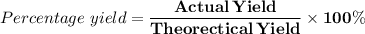
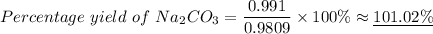
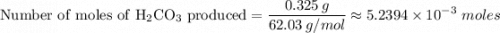
 Na₂CO₃(s) + H₂CO₃(g)2 moles of NaHCO₃ produces 1 mole of H₂CO₃
Na₂CO₃(s) + H₂CO₃(g)2 moles of NaHCO₃ produces 1 mole of H₂CO₃