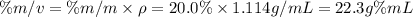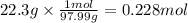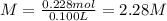Chemistry, 18.07.2020 04:01, lollollollollol1
A 20.0 % by mass solution of phosphoric acid (H 3PO 4) in water has a density of 1.114 g/mL at 20°C. What is the molarity of this solution?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:50, zaleemawhite
Significant figures are digits read directly from the measuring instrument plus one more digit, which is __ by the observer.
Answers: 2

Chemistry, 22.06.2019 08:30, ayaanwaseem
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2

Chemistry, 22.06.2019 11:00, 21villalobosjabez
Which type of fossil does this image depict?
Answers: 1

Chemistry, 22.06.2019 11:30, ansuaprajita1506
Voltaic cells produce a positive overall charge. what does this indicate? a. the reaction is likely to be endothermic. b. the reaction is spontaneous. c. the reaction is not likely to occur. d. the reaction is not spontaneous.
Answers: 3
Do you know the correct answer?
A 20.0 % by mass solution of phosphoric acid (H 3PO 4) in water has a density of 1.114 g/mL at 20°C....
Questions in other subjects:

History, 04.08.2019 03:40

Social Studies, 04.08.2019 03:40


History, 04.08.2019 03:40

Social Studies, 04.08.2019 03:40

Physics, 04.08.2019 03:40