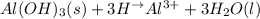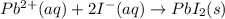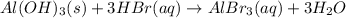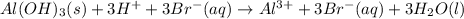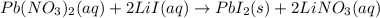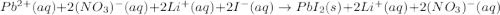Chemistry, 17.07.2020 04:01, chaitanyac90
Write a balanced molecular and net ionic equation for the following reactions
a. insoluble aluminum hydroxide reacts with hydrobromic acid
b. Lead (I) nitrate reacts with lithium iodide to produce a precipitate and a soluble compound.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 21:30, sarah192002
What is happening when the water inside a kettle heats up and begins to boil
Answers: 1

Chemistry, 23.06.2019 01:20, michellectucker1982
Use the de broglie's wave equation to find the wavelength of an electron moving at 7.3 × 106 m/s. show your work. note: h = plank's constant (6.62607 x 10-34 j s)
Answers: 1

Chemistry, 23.06.2019 01:30, jarteria0
Some molecular compounds, such as hcl, ionize completely in solution. for molecular compounds such as h2co3, most molecules do not ionize in solution. which describes the properties of these two solutes? a. hcl and h2co3 have the same effect on the properties of the solution. b. hcl raises the freezing point of water more than h2co3 does. c. hcl raises the boiling point of water more than h2co3 does.
Answers: 2
Do you know the correct answer?
Write a balanced molecular and net ionic equation for the following reactions
a. insoluble aluminum...
Questions in other subjects:










Geography, 06.05.2020 18:09