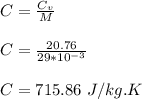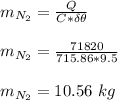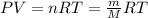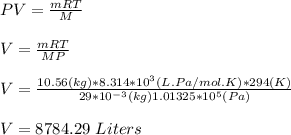Chemistry, 15.07.2020 20:01, xXCoryxKenshinXx
g compute the specific heat capacity at constant volume of nitrogen gas. the molar mass of N2 is 29.0 You warm 1.8 kg ov water at a constant volume from 21 C to 30.5 C in a kettle

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, lindseyklewis1p56uvi
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 22.06.2019 09:30, raizagisselle1694
Mike and mitchell decide to have a foot race. they mark off a stretch of 100 yards, and recruit cindy to work the stopwatch. after running the race and looking at the results, cindy declared that mitchell was the fastest. so how did the boys times compare?
Answers: 3

Do you know the correct answer?
g compute the specific heat capacity at constant volume of nitrogen gas. the molar mass of N2 is 29....
Questions in other subjects:

Arts, 04.11.2019 23:31

Chemistry, 04.11.2019 23:31

Computers and Technology, 04.11.2019 23:31

Mathematics, 04.11.2019 23:31


History, 04.11.2019 23:31


Health, 04.11.2019 23:31

English, 04.11.2019 23:31