Chemistry, 14.07.2020 01:01, gissellesolorza
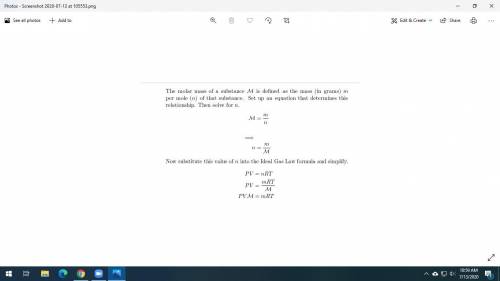
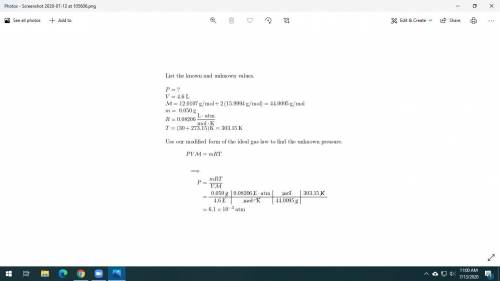
Dry ice is solid carbon dioxide. A 0.050-g sample of dry ice is placed in an evacuated 4.6-L vessel at 30 °C. Calculate the pressure inside the vessel after all the dry ice has been converted to CO2 gas.

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 11:00, snowprincess99447
Which statement is true about hcl? (5 points) select one: a. it is a salt because it increases the concentration of metallic ions. b. it is a salt because it is formed by the reaction of an acid and a base. c. it is an acid because it increases the concentration of hydroxyl ions. d. it is an acid because it increases the concentration of hydronium ions.
Answers: 1

Chemistry, 22.06.2019 12:00, winterblanco
What is the lowest number energy level where a d sublevel is found
Answers: 1
Do you know the correct answer?
Dry ice is solid carbon dioxide. A 0.050-g sample of dry ice is placed in an evacuated 4.6-L vessel...
Questions in other subjects:



Health, 22.04.2021 20:20

Biology, 22.04.2021 20:20




Mathematics, 22.04.2021 20:20


History, 22.04.2021 20:20