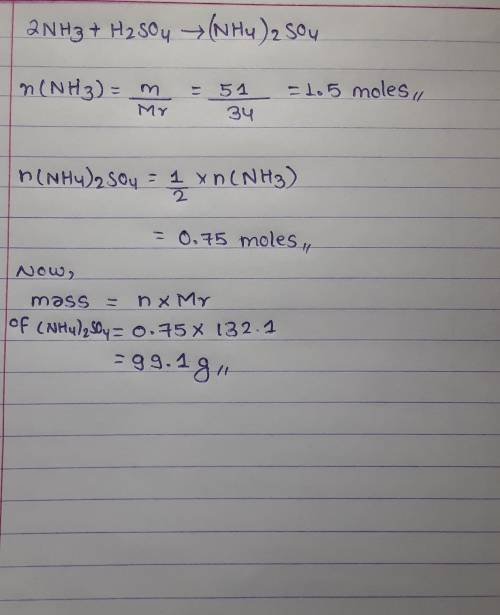Chemistry, 14.07.2020 01:01, saraaaaaaaa20
calculate the mass of ammonium sulfate that can be made from 51 grams of ammonia. 2NH₃ + H₂SO₄ → (NH₄)₂SO₄ (keeping the mole concept in mind)

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:40, trinityanne1738
Asolid that forms and separates from a liquid mixture is called
Answers: 2


Chemistry, 22.06.2019 17:40, aaliyahthomas37
Which statement about hf is true? it is zero for any compound in its standard state. it is positive when the bonds of the product store more energy than those of the reactants. it is negative when a compound forms from elements in their standard states. it is zero for any element that is in the liquid state.
Answers: 1

Chemistry, 22.06.2019 18:50, emily9656
Which of the following is a conclusion that resulted from ernest rutherford’s scattering experiment? (will mark brainliest) a. the nucleus is negatively charged b. the atom is a dense solid and is indivisible c. the mass is conserved when atoms react chemically d. the nucleus is very small and the atom is mostly empty space
Answers: 3
Do you know the correct answer?
calculate the mass of ammonium sulfate that can be made from 51 grams of ammonia. 2NH₃ + H₂SO₄ → (NH...
Questions in other subjects:

Mathematics, 16.04.2021 14:00

English, 16.04.2021 14:00


Mathematics, 16.04.2021 14:00

Chemistry, 16.04.2021 14:00



Mathematics, 16.04.2021 14:00

Mathematics, 16.04.2021 14:00

Social Studies, 16.04.2021 14:00