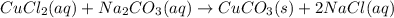Mixing which 0.1 M aqueous solutions results in formation of a colored precipitate? A) BaCl2 and CH3COOH (B) BaCl2 and Na2CO3 (C) CuCl2 and CH3COOH (D) CuCl2 and Na2CO3 The correct answer is D but please explain I'm confused. I know that the product has to be insoluble to form a precipitate, but how do you know if it'll be colored?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:30, cheyennecarrillo14
If you add 5.00 ml of 0.100 m sodium hydroxide to 50.0 ml of acetate buffer that is 0.100 m in both acetic acid and sodium acetate, what is the ph of the resulting solution? acetic acid: ka = 1.8. x 10-5
Answers: 1

Chemistry, 22.06.2019 18:40, bananaslada
What is the binding energy of a nucleus that has a mass defect of 5.81*10-^29 kg a 5.23*10-^12 j b 3.15* 10^12 j c 1.57*10-3 j d 9.44*10^20 j
Answers: 1

Chemistry, 23.06.2019 02:00, raulflores01
Which best describes the present-day universe? opaque, expanding very slowly, stars produce heavy elements transparent, expanding at an accelerated rate, stars produce heavy elements opaque, expanding at an accelerated rate, stars produce only hydrogen and helium transparent, expanding very slowly, stars produce only hydrogen and helium
Answers: 1

Chemistry, 23.06.2019 05:30, cluchmasters5634
Scientist think that animals with remarkably similar embryo development probably shared a common ancestor
Answers: 1
Do you know the correct answer?
Mixing which 0.1 M aqueous solutions results in formation of a colored precipitate? A) BaCl2 and CH3...
Questions in other subjects:


English, 06.04.2020 22:38

Medicine, 06.04.2020 22:38

Geography, 06.04.2020 22:38



Physics, 06.04.2020 22:38