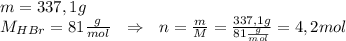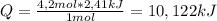Chemistry, 21.01.2020 14:31, naomicervero
How much heat energy is required to melt 337.1 g of hbr? the molar heat of fusion of hbr is 2.41 kj/mol.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, jbarbie3
12. complete each of the following word equations for synthesis reactions. a. sodium + oxygen -> b. magnesium + fluorine -> 13. complete and balance the equations for the decomposition reactions. a. hgo -> [with the triangle heat symbol above the arrow] b. h2o(l) -> [with "electricity" written above the arrow]
Answers: 1


Chemistry, 22.06.2019 07:30, deidaraXneji
Using data from seismic waves, geologists have learned that earth’s interior is made up of several
Answers: 3

Chemistry, 22.06.2019 09:40, kolibeilfuss
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3
Do you know the correct answer?
How much heat energy is required to melt 337.1 g of hbr? the molar heat of fusion of hbr is 2.41 kj...
Questions in other subjects:



English, 13.01.2021 18:30







Computers and Technology, 13.01.2021 18:30