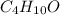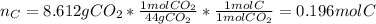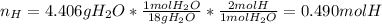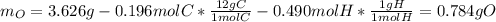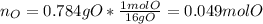Chemistry, 09.07.2020 02:01, germainenez3288
The compound known as diethyl ether, commonly referred to as ether, contains carbon, hydrogen, and oxygen. A 3.626 g sample of ether was combusted in an oxygen rich environment to produce 8.612 g of CO2(g) and 4.406 g of H2O(g). Insert subscripts to complete the empirical formula of ether.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:00, asims13
Which element in the third period would you expect to have the larger atomic radius, sodium (na) or sulfur (s)? a. sodium, because it has a higher effective nuclear charge attracting electrons in fewer energy levels. b. sodium, because it has fewer protons attracting electrons in the same energy levels. c. sulfur, because it has more protons attracting electrons in more energy levels. d. sulfur, because it has a higher effective nuclear charge attracting electrons in the same energy levels.
Answers: 2

Chemistry, 21.06.2019 19:00, cutebab4786
Consider the point on the plot where 10.0 g of naoh have been added. what amount of naoh, in moles, has been added? 0.308 mol fecl3 initially present
Answers: 1

Chemistry, 22.06.2019 04:50, aletadaboss
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1

Chemistry, 22.06.2019 13:10, kellinvagneur
Which electron configuration represents the electrons in an atom of sodium in the ground state at stp
Answers: 1
Do you know the correct answer?
The compound known as diethyl ether, commonly referred to as ether, contains carbon, hydrogen, and o...
Questions in other subjects:

Social Studies, 15.09.2020 02:01

Social Studies, 15.09.2020 02:01

Mathematics, 15.09.2020 02:01

Mathematics, 15.09.2020 02:01

Mathematics, 15.09.2020 02:01

Mathematics, 15.09.2020 02:01

Mathematics, 15.09.2020 02:01

English, 15.09.2020 03:01

Mathematics, 15.09.2020 03:01

Mathematics, 15.09.2020 03:01