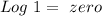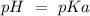Chemistry, 09.07.2020 01:01, jybuccaneers2022
50 mL of 0.1 M acetic acid is mixed with 50 mL of 0.1 M sodium acetate (the conjugate base). The Ka of acetic acid is approximately 1. 74 X 10 -5. What is the pH of the resulting solution?

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 16:00, rorymartin04
No copying 15 pts how does a free-body diagram tell you about the net force on an object?
Answers: 2

Chemistry, 22.06.2019 16:10, 00015746
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2
Do you know the correct answer?
50 mL of 0.1 M acetic acid is mixed with 50 mL of 0.1 M sodium acetate (the conjugate base). The Ka...
Questions in other subjects:


Health, 07.10.2019 19:30

English, 07.10.2019 19:30


English, 07.10.2019 19:30






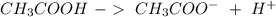
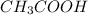 ) and a base (
) and a base (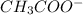 ). Therefore we can write the henderson-hasselbach reaction:
). Therefore we can write the henderson-hasselbach reaction:![pH~=~pKa+Log\frac{[CH_3COO^-]}{[CH_3COOH]}](/tpl/images/0703/3490/99062.png)
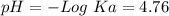
![[CH_3COO^-]=[CH_3COOH]](/tpl/images/0703/3490/ee54c.png)
![\frac{[CH_3COO^-]}{[CH_3COOH]}~=~1](/tpl/images/0703/3490/6e489.png)