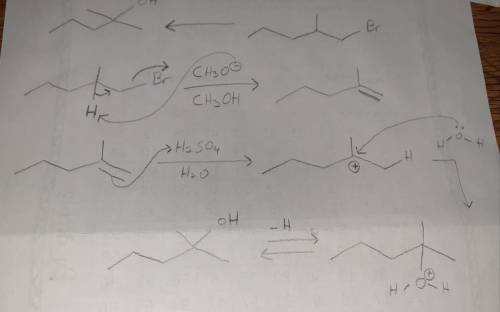
Design a Synthesis of 2-methyl-2-pentanol from 1-bromo-2-methylpentane Part 1: Choose the best option for the immediate precursor to the target molecule. An alkene that has greater substitution on one end of the double bond permits you to control the regiochemistry, placing the OH group selectively on one of the two double bond carbons. This alkene is the best choice, considering the starting material that you have available. Part 2: Choose the best option for the precursor needed to make the alkene. Alkenes can be made in good yield by elimination of HBr from an alkyl halide. Whenever possible, use an alkyl halide that can only make a single alkene by HBr elimination. Part 3: Here is an overview of your retrosynthesis (there is no work, this is just a recap). Part 4 out of 6 Choose the most appropriate reagent(s) for the first step of the synthesis.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:00, bobbycisar1205
How does a hydroelectric power plant converts energy into energy.
Answers: 1



Chemistry, 22.06.2019 19:40, jholland03
What type of electromagnetic waves does the human eye see as the colors red blue or green a visible light waves b radio waves c infrared waves d microwaves
Answers: 1
Do you know the correct answer?
Design a Synthesis of 2-methyl-2-pentanol from 1-bromo-2-methylpentane Part 1: Choose the best optio...
Questions in other subjects:



History, 27.06.2019 16:00

Biology, 27.06.2019 16:00


Mathematics, 27.06.2019 16:10

Mathematics, 27.06.2019 16:10



Mathematics, 27.06.2019 16:10