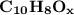Chemistry, 07.07.2020 21:01, guapotaco8102
4.50 g of a certain Compound X, known to be made of carbon, hydrogen and perhaps oxygen, and to have a molecular molar mass of 128. g/mol, is burned completely in excess oxygen, and the mass of the products carefully measured: product carbon dioxide water mass 15.47 g 2.53 g Use this information to find the molecular formula of X.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 15:00, Dreambig85
Use examples from the article to explain one positive and one negative effect that chemistry has had on society
Answers: 2

Chemistry, 21.06.2019 19:30, maddietomlinson113
If the element whose electric configuration ends in the d sublevel, the element is calssified as? a. inner transition b. noble gases c. representative d. transition
Answers: 2

Chemistry, 22.06.2019 13:30, yasiroarafat12
How many moles is 14.5 cm^3 of platinum? the density of platinum is 21.45 g/cm^3.
Answers: 1

Chemistry, 23.06.2019 00:30, zaniathomasel
What is bromine+calcium iodide--> calcium bromide +iodine balanced
Answers: 1
Do you know the correct answer?
4.50 g of a certain Compound X, known to be made of carbon, hydrogen and perhaps oxygen, and to have...
Questions in other subjects:

History, 29.05.2020 22:00

Mathematics, 29.05.2020 22:00

Chemistry, 29.05.2020 22:00

Social Studies, 29.05.2020 22:00

History, 29.05.2020 22:00

Health, 29.05.2020 22:00




Chemistry, 29.05.2020 22:00


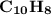 ( Naphthalene )
( Naphthalene )