
Chemistry, 04.07.2020 14:01, aeshaalhemri
A weather balloon is inflated to a volume of 27.6 L at a pressure of 755 mmHg and a temperature of 29.9 ∘C. The balloon rises in the atmosphere to an altitude where the pressure is 385 mmHg and the temperature is -14.1 ∘C. Assuming the balloon can freely expand, calculate the volume of the balloon at this altitude.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:00, melidacampos12
Substance x has a fixed volume, and the attraction between its particles is strong .substance y had widely spread out particles and can be compressed what can most likely be concluded about these substances
Answers: 2


Chemistry, 22.06.2019 07:00, vivianni0727p1y30v
How heavy is thanos? a) 3000 lbs b) all of it c) the price of tea in china d) heavy enough
Answers: 2

Chemistry, 22.06.2019 19:00, cindyroxana229
Nan element’s square on the periodic table, the number with the greatest numerical value represents the
Answers: 3
Do you know the correct answer?
A weather balloon is inflated to a volume of 27.6 L at a pressure of 755 mmHg and a temperature of 2...
Questions in other subjects:


History, 05.12.2019 06:31



English, 05.12.2019 06:31

English, 05.12.2019 06:31

History, 05.12.2019 06:31




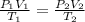
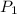 = initial pressure of gas = 755 mm Hg
= initial pressure of gas = 755 mm Hg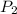 = final pressure of gas (at STP) = 385 mm Hg
= final pressure of gas (at STP) = 385 mm Hg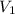 = initial volume of gas = 27.6 L
= initial volume of gas = 27.6 L = final volume of gas = ?
= final volume of gas = ? = initial temperature of gas =
= initial temperature of gas = 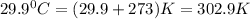
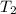 = final temperature of gas =
= final temperature of gas = 






