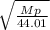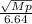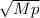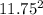When 3.51 g of phosphorus was burned in chlorine, the product was a phosphorus chloride. Its vapor took 1.77 times as long to effuse as the same amount of CO2 under the same conditions of temperature and pressure. What is the molar mass of the phosphorus chloride

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:20, lex68259100
Which formula can be used to calculate the molar mass of ammonia (nh3)? molar mass of n + molar mass of h 3 × molar mass of n + molar mass of h molar mass of n + 3 × molar mass of h 3 × molar mass of n + 3 × molar mass of h
Answers: 1

Chemistry, 22.06.2019 10:30, Wookas8355
Great amounts of electromagnetic energy from our sun and other bodies in space travel through space. which is a logical conclusion about these electromagnetic waves? their energy must be very their frequency must be very low these waves can travel without a medium they only travel through a vacuum of space
Answers: 2


Chemistry, 22.06.2019 17:40, adantrujillo1234
Areaction in which products can react to re-form reactants is
Answers: 1
Do you know the correct answer?
When 3.51 g of phosphorus was burned in chlorine, the product was a phosphorus chloride. Its vapor t...
Questions in other subjects:


Mathematics, 09.12.2020 07:50


Chemistry, 09.12.2020 07:50



Mathematics, 09.12.2020 07:50

Mathematics, 09.12.2020 07:50


Mathematics, 09.12.2020 07:50


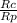 =
= 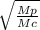
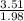 =
=